 **PLEASE SEE UPDATES BELOW**
**PLEASE SEE UPDATES BELOW**
It is commonly believed that distinct mini-networks of neurons, firing together, may be the means by which memories and other conceptual encoding requirements are handled in the brain. However, it is only recently that we have had the tools available to directly test the sufficiency of such a mechanism. Today, a new study in Nature from the lab of Susumu Tonegawa documents the ability to use light as a means to activate distinct subsets of neurons responsible for the encoding of fear memories.
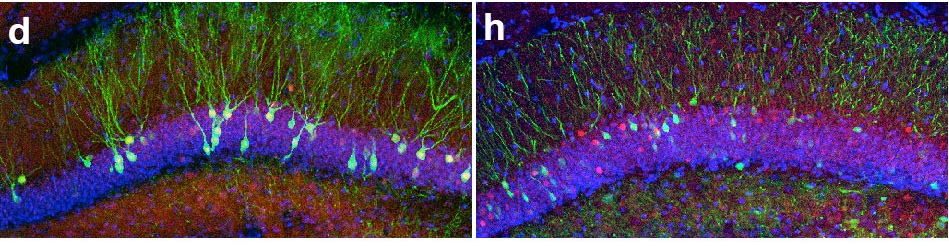
To gain access to cells active during memory encoding, the authors used a clever genetic system in which the promoter of a gene known to be expressed after strong neuronal activation (c-fos) was engineered to drive expression of channelrhodopsin, a light-activated channel able to induce neuronal firing after exposure to blue light. For temporal specificity, the entire system was also under the control of an inducible element, which only allowed the engineered c-fos promoter to operate in the absence of a chemical (doxycycline, or dox,) usually added to the drinking water of the animals. So, for neurons to eventually express channelrhodopsin, they would have to be dox-free and active. There are a variety of controls in the paper to demonstrate the legitimacy of the system and in fact, one example is in the figure above. On the left is the dentate gyrus region of the hippocampus with cells expressing channelrhodopsin tagged with eGFP (green). Upon light exposure, these cells became active and expressed c-fos (immunohistochemical staining, in red.) Although there is additional red that is not overlapping with green signals, almost all green cells contain overlapping red (c-fos) staining. On the right, the dentate gyrus of another animal is shown with cells expressing eGFP only. There are no neurons that should be responding to light and therefore, red c-fos staining simply reflects randomly active neurons. These red cells do not overlap with green cells at any rate greater than chance, demonstrating the strong relationship between neurons expressing channelrhodopsin and c-fos-active neurons (the former should mostly all be a subset of the latter in the presence of light!)
So now the authors had a system in which they could target active neurons and gain access to them again later. In the key experiment, animals were habituated in Context A, with baseline levels of fear quantified by the amount of time the animal spends “freezing.” Freezing is a good measure of fear in rodents because it is an instinctual reaction in the face of vision-driven predators, like birds of prey, looking for movement to cue them on to a potential meal. Of course, animals expressed little freezing behavior in Context A since there was no obvious threat being presented. Animals were then taken off dox and placed in Context B, where they experienced footshocks, presumably triggering the encoding of fear memories related to this negative experience. Although fear memories can be complex, contextual aspects are known to be documented by the hippocampus, one of the areas of the brain encoding information related to location. Thus, in Context B, these hippocampal dentate gyrus cells were becoming strongly activated in an attempt to encode everything about this potentially dangerous context for future reference. And since dox was no longer in the system, these active cells were happily producing channelrhodopsin. After contextual fear training, dox was reintroduced to stop the expression of channelrhodopsin in additional cells unrelated to contextual fear memory simply through random experience and spontaneous activity. Then came the big test. Would the light-driven activation of those cells firing away during the contextual fear encoding be sufficient to induce the fearful behavior of freezing outside the fearful context? Animals were returned to the safe confines of Context A and were allowed to explore in the presence or absence of light activation of channelrhodopsin-expressing cells. Light was introduced via the surgical implantation of a small fiber optic cable directly above the dentate gyrus. Indeed, light-activation of fear-associated, Context B-linked cells induced freezing in neutral Context A, while no freezing was observed in the absence of light exposure. So essentially, the animals were uncontrollably expressing fearful behaviors simply because circuits created to respond to an experienced dangerous context were being artificially activated, bypassing executive control and simply short-circuiting the initiation of freezing behavioral programs. It’s hard not to think that’s pretty cool.
But despite demonstrating the sufficiency of these microcircuits, or “memory engrams,” to invoke fear response out of context, what would be really nice is also demonstrating necessity. In fact, this was raised during the review process. A subset of the reviewers (and me) would have liked to see the authors disrupt this memory engram in the appropriate context, say by expressing a different light-activated channel in the participating neurons to shut the cells off, and document the result. In this scenario, although the animals would usually invoke freezing behaviors naturally in the dangerous Context B, if the same cells “remembering” Context B were inhibited, then the rodent may not express fear. It sounds great in principle, but the authors basically argued that fear memories are much more complicated than that. Removing only one trigger may not be enough, or perhaps the engram could be more complicated than what they could target genetically for inactivation, so a negative result may not be interpretable. After rounds of discussion and additional controls, both the editors and (most of) the reviewers were convinced that pursuing the question of necessity was much more complex, may not lead to a clean resolution and would unduly delay publication of this exciting result that already demonstrated sufficiency of the engram to induce fear.
This was a good thing for the authors for a number of reasons, not least because after we accepted their manuscript, they were made aware of a similar paper close to publication in Science asking similar types of questions, but using distinct methods and strategies. Now usually we are moderately speedy with preparing papers for online publication (perhaps 3-4 weeks,) but the authors were concerned enough about being “scooped” that we decided to “fast-track” the production of the paper in order for it to appear at the same time as the competing work in Science. It took about a week after receiving the final version from the authors for our production team to create the “near-final version” of the manuscript, the one that is currently linked-to above. Once it was clear that we were on track and ready for online publication, the only thing left to do was to coordinate publication with Science. Simple communications between Nature‘s press office and their counter-part at Science confirmed the presence of a similar paper and determined that Science would be releasing their paper today. So we did the same, with the same exact release time. This paper, “Generation of a Synthetic Memory Trace,” from the laboratory of Mark Mayford can be found here. I would’ve written that one up too, but it’s already past 16:00, I’ve got even more awesome papers to read and I need more coffee.
03/23/12 UPDATE: Your prayers have been answered because Jason Snyder of the Functional Neurogenesis blog has written up more on the complementary paper from the Mayford group. Even more coverage from science writer Ed Yong as well as MIT News.
03/23/12 UPDATE #2: As mentioned in the comments below, Hysell Oviedo remembers the original public demonstration of using this methodological strategy to re-activate specific memory traces using optogenetics. This occurred in a presentation at the Society for Neuroscience meeting in the fall of 2009, from the laboratory of Michael Häusser. Dr. Häusser was kind enough to provide us with the original press release from SfN here [PDF]. This is interesting, given the current discussion over the future of science communication and publications. Under a different scientific culture and data presentation system, this original abstract would have been discussed and vetted for years, inspiring the authors to improve the work in a public manner, with no question as to precedence. Under our current system, unless you were at SfN 09 (and even if you were,) it is likely you never saw this work at all. At least until now…

@hysell has pointed out on twitter that there was a 2009 SFN abstract describing a similar study, blogged about here: https://brainwindows.wordpress.com/2009/09/18/optogenetic-induction-of-memory-recall/
The Rizzi & Häusser SfN abstract was relayed by a few blogs, and by Nature News at the time: https://www.nature.com/news/2009/091020/full/news.2009.1016.html. So it was a bit more public that the last phrase would lead to think. The question remains though, why this work was not published until now.
Thanks for the link, so that even makes the point over receiving intellectual credit at major career transitions outside of publications even that much stronger. I’m not saying a news story on nature.com should get you tenure, but the sum total of all such coverage, capturing the imagination of those inside and outside the field, should count for something.
I agree with Leterrier: The Hausser SFN abstract was the single most-talked-about SFN abstract that I can recall…although at the time —as with most posters— the controls were not comprehensive. This project was something that many groups (I can think of at least 4) have been working very hard on for the last 3-5 years, and credit should be given to all groups that pushed this forward and actually implemented this tool. Necessity is a very interesting question, and this leaves us some work to be done…This is definitely a major advance that I have personally been eagerly waiting for since the previous Mayford paper was published.
So my pseudo-trolling has raised the important point: that many in the field (and outside) did know this work and the potential project quite well. Just adding more fodder to the discussion of how intellectual property, credit and primacy are established. Häusser’s lab was public with their progress and data. Tonegawa’s lab published it. Imagine if all four labs that Kay mentions were presenting progress and data over the past 3-5 years. That could have resulted in very fruitful discussions and explorations of the topic. Science in the open is fascinating and only dangerous if we make it so. Perhaps certain groups could have provided feedback and discussion, but focused more on the “necessity” experiments, while Tonegawa’s lab championed the sufficiency ones, etc, etc… In a financial climate like the one we are currently on, why double-up on experiments when communication can better organize the science and the distribution of grant resources? Much more efficiency can be squeezed from the resource dollar.
But sadly, it is still extremely dangerous to be open.
Indeed, the community felt this experiment was low-hanging fruit just begging to be plucked. Nevertheless, this episode will boost a trend we all complain about: people NOT showing unpublished data at scientific meetings. It is not uncommon for posters to be pulled at the last minute because the paper hasn’t been submitted. It is not uncommon for big labs to browse posters from small labs for ideas they can do faster and sexier. And the more tech-driven the field the worse it gets. This in part has driven the explosion of small, invite-only meetings. They create an environment of accountability: I know the handful of people who saw my unpublished work, so I’ll be watching you closely… We’re clearly en route to (if not there already) a winner-take-all system in scientific research. Funding agencies and the scientific community need to think hard about whether this is a good direction because resources are scarce, and if so implement it explicitly.