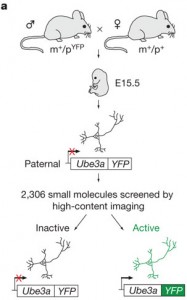
Here’s one that first appeared online at the end of last year by Benjamin Philpot, Bryan Roth and Mark Zylka about a finding that could lead to a therapy for Angelman Syndrome. Angelman syndrome is a rare neurodevelopmental disorder affecting 1 in 15,000 live births and is characterized by developmental delay, lack of speech, seizures, and motor difficulties. There are no therapies available for core symptoms and individuals generally require care throughout life. Autism is often diagnosed in Angelman Syndrome individuals, and the same genomic region has been fingered as a culprit in both disorders. Angelman Syndrome is most commonly caused by deletion of a region on the maternal copy of chromosome 15 containing the gene UBE3A, conversely, some forms of autism may also be caused by duplication of this region.
Although we all possess two copies of UBE3A, only the one inherited from the mother is active. Normally, the paternal copy is epigenetically silenced. This means that in Angelman Syndrome there is no functioning copy at all, which has consequences for multiple signaling pathways and brain circuits. The authors of this paper set out to find a workaround: something that could activate the intact, but dormant, paternal copy of UBE3A. They made a reporter assay from neurons of mice expressing fluorescent paternal UBE3A protein, and performed a large-scale drug screen, testing over 2000 compounds for ones that would activate paternal Ube3A. None of the most likely suspects worked, but an unlikely class of drugs, topoisomerase inhibitors, did so reliably. Even better, one of the best (topotecan) is already an FDA-approved cancer drug.