We’ve known for over a century that sensory cortex is arranged in distinct layers, each containing a different make up of neuronal types and projection patterns, but we don’t actually know that much about the actual computations performed in each layer. Today a paper from Massimo Scanziani’s lab takes a big step towards cracking the function of the bottom layer (layer 6) in mice. Layer 6 neurons project both to upper cortical layers and to the lateral geniculate nucleus in the thalamus, which itself is the primary input to cortex, and so are primed to play a large modulatory role. Using a monumental combination of optogenetics, intracellular recording, and behavioral testing, the paper convincingly makes the case that layer 6 controls the gain of visual responses of upper layer neurons (i.e. changes the size of their responses without altering their selectivity). Gain control is a fundamental computation in cortex, and has been invoked as a mechanism for attention, perception, spatial processing, and more. The cellular mechanism here is worked out in primary visual cortex, but it could potentially operate throughout layered cortex.
Monthly Archives: February 2012
Lost in Translation — Chasing the Roots of Conditioned Fear Research
I’m currently attending the Winter Conference on Neural Plasticity in lovely St. Kitts & Nevis and I’ll be tweeting when I can from #wcnp12 when the Internet access in the room decides to cooperate.
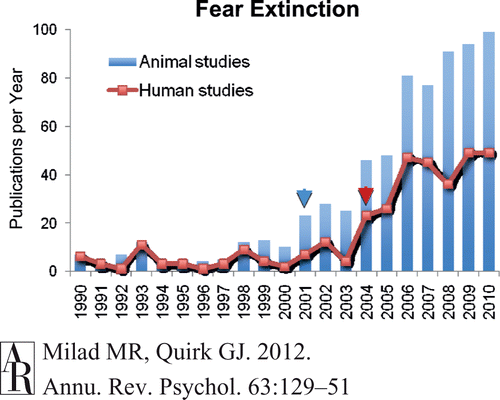
Today’s opening session at the meeting was a historical perspective on selected topics in neural plasticity. I thought I’d share an interesting piece of history about one topic that has exploded in terms of research output over the last 20 years: conditioned fear. Michael Fanselow gave the lecture on the history of fear research and focused on the era prior to the exponential growth of the literature, sticking to 1920-1980. Here’s a graph from a very recent review simply noting the number of “fear extinction” papers in the literature (one small sub-field in this topic,) just to give you a sense of how rapidly this field has grown:
I’ll do may best to channel Dr. Fanselow with the next few paragraphs:
Telepathy? I think not
There is just something about neural decoding that captures the imagination. Scientists “reading out brain activity” to infer what someone was seeing or doing sounds like the stuff of science fiction. But in practice, with the right dataset and right computer algorithm, it can be done – providing the question you are trying to query the brain is simple enough. But no matter how simple the question, with every paper comes an orgy of stories in the mainstream press about how scientists can eavesdrop on your thoughts or even engage in electronic telepathy. Thereby infuriating scientists and science journalists in droves, sometimes detracting from some very cool work.
Today I’m going back a few years to a paper that typifies this effect, a study from Jack Gallant‘s lab about a model for decoding natural images from fMRI activity in early visual cortex.
Awakening dormant genes with cancer drugs
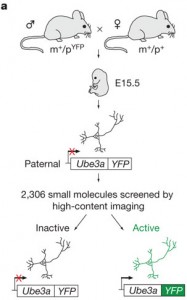
Here’s one that first appeared online at the end of last year by Benjamin Philpot, Bryan Roth and Mark Zylka about a finding that could lead to a therapy for Angelman Syndrome. Angelman syndrome is a rare neurodevelopmental disorder affecting 1 in 15,000 live births and is characterized by developmental delay, lack of speech, seizures, and motor difficulties. There are no therapies available for core symptoms and individuals generally require care throughout life. Autism is often diagnosed in Angelman Syndrome individuals, and the same genomic region has been fingered as a culprit in both disorders. Angelman Syndrome is most commonly caused by deletion of a region on the maternal copy of chromosome 15 containing the gene UBE3A, conversely, some forms of autism may also be caused by duplication of this region.
Although we all possess two copies of UBE3A, only the one inherited from the mother is active. Normally, the paternal copy is epigenetically silenced. This means that in Angelman Syndrome there is no functioning copy at all, which has consequences for multiple signaling pathways and brain circuits. The authors of this paper set out to find a workaround: something that could activate the intact, but dormant, paternal copy of UBE3A. They made a reporter assay from neurons of mice expressing fluorescent paternal UBE3A protein, and performed a large-scale drug screen, testing over 2000 compounds for ones that would activate paternal Ube3A. None of the most likely suspects worked, but an unlikely class of drugs, topoisomerase inhibitors, did so reliably. Even better, one of the best (topotecan) is already an FDA-approved cancer drug.