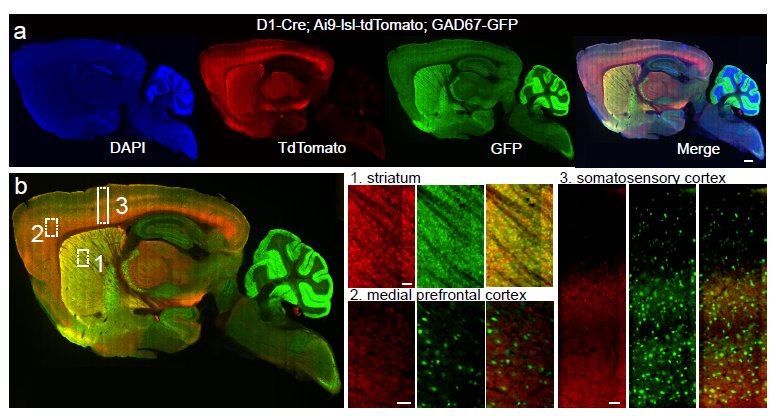
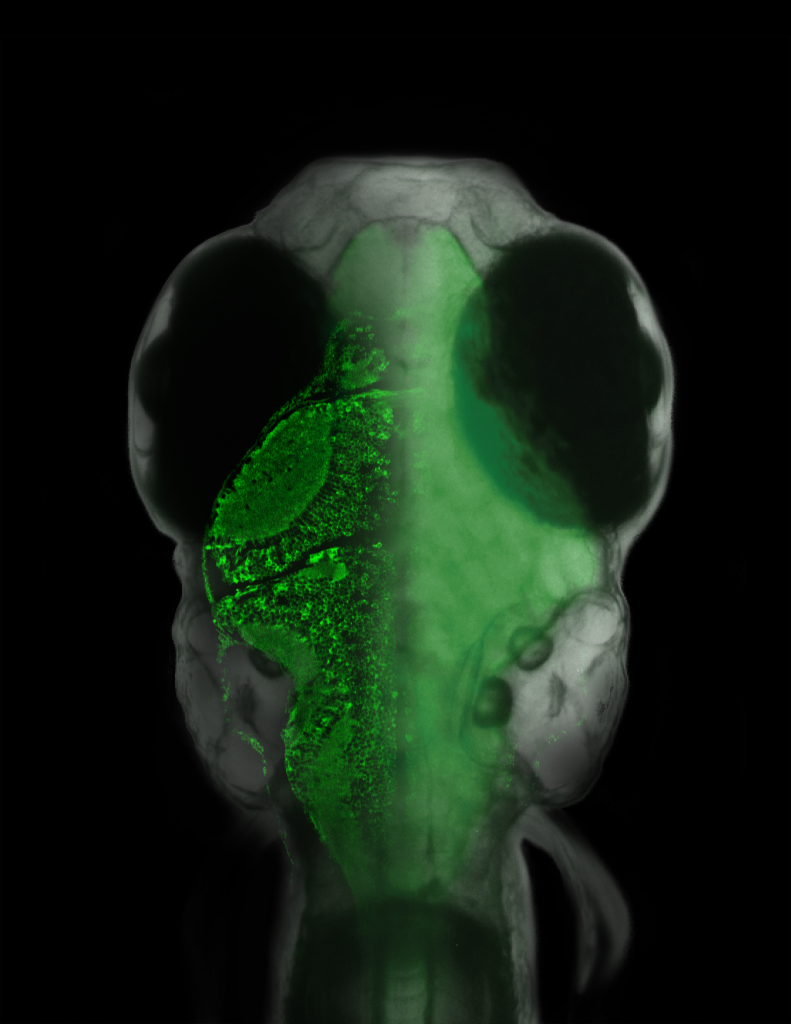
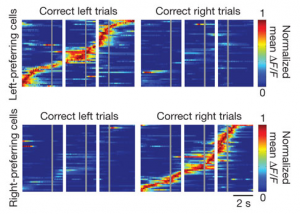
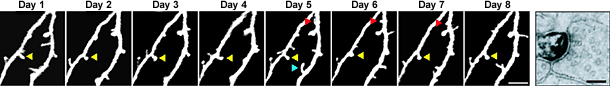
It really is an embarrassment of riches here at Nature these days, what with so many excellent neuroscience-related studies emerging. Just in the last couple of weeks, we’ve had the following studies:
- Covert learning by a basal ganglia circuit, despite no participating in the behavioral practice.
- Reach and grasp by people with tetraplegia using a neurally-controlled robotic arm.
- A non-transcriptional circadian cycle conserved across all domains of life.
- Identification of mechanisms linking cerebro-vascular integrity to neurodegeneration.
- Metabolic coupling of glia with axons in the long-term maintenance of axonal integrity.
So really, a lot to write about from a science perspective. However, this blog is dedicated to bringing you the editorial back-story, so I wanted to touch on yet another interesting study, published in print today. This new paper offers an opportunity to discuss an important editorial issue: the manuscript appeal process. For more details, you can always read the appropriate section in our guide to authors. But it’s often helpful to follow a particular [successful] example in order to illustrate the process. Continue reading