Genetic clues to what makes parasitic worms different from free-living worms are reported in a paper published online this week in Nature Genetics. Groups led by Mark Viney, Matthew Berriman and Taisei Kikuchi carried out the sequencing and assembly of genomes from six nematode species from the clade that includes the human parasitic roundworm Strongyloides stercoralis. We asked one of the authors, Professor Mark Viney of the University of Bristol, to tell us a little bit about the study.
Although the genomes of several parasitic worm species have been published to date, Strongyloides represents a unique opportunity to learn some of the general rules of being a parasitic worm. According to Mark Viney, “what makes Strongyloides so special is that this clade contains parasites, facultative parasites and free-living species that are all close relatives. This gives us real power to our analysis. Our work will be used by the international research community who work on these globally important parasites of people and other animals.”
S. stercoralis infects approximately 30-100 million people worldwide and causes a wide range of symptoms. Closely related species in the clade Strongyloides include both free-living and parasitic species that infect a wide range of hosts. In parasitic species, generations alternate between parasitic and free-living, resulting in genetically identical females with starkly different lifestyles.
The authors first compared the genomes of free-living and parasitic species to identify genes specific to the parasites. They found that acquisition of 1,075 gene families was associated with the evolution of parasitism and parasitism was associated with greater expansion of genes and gene families overall.
When asked what the most unexpected aspect of the study was, Professor Viney said, “I think the really surprising thing that we found was just how largely expanded some gene families were in the parasitic species. This is quite unprecedented in the nematodes.” The authors also found that most parasitism-related genes were located in genomic clusters. “The important thing about these clusters is that nothing like this has ever been seen before in parasitic worms and it certainly speaks to the possible importance of these in their evolution of parasitism,” said Professor Viney.
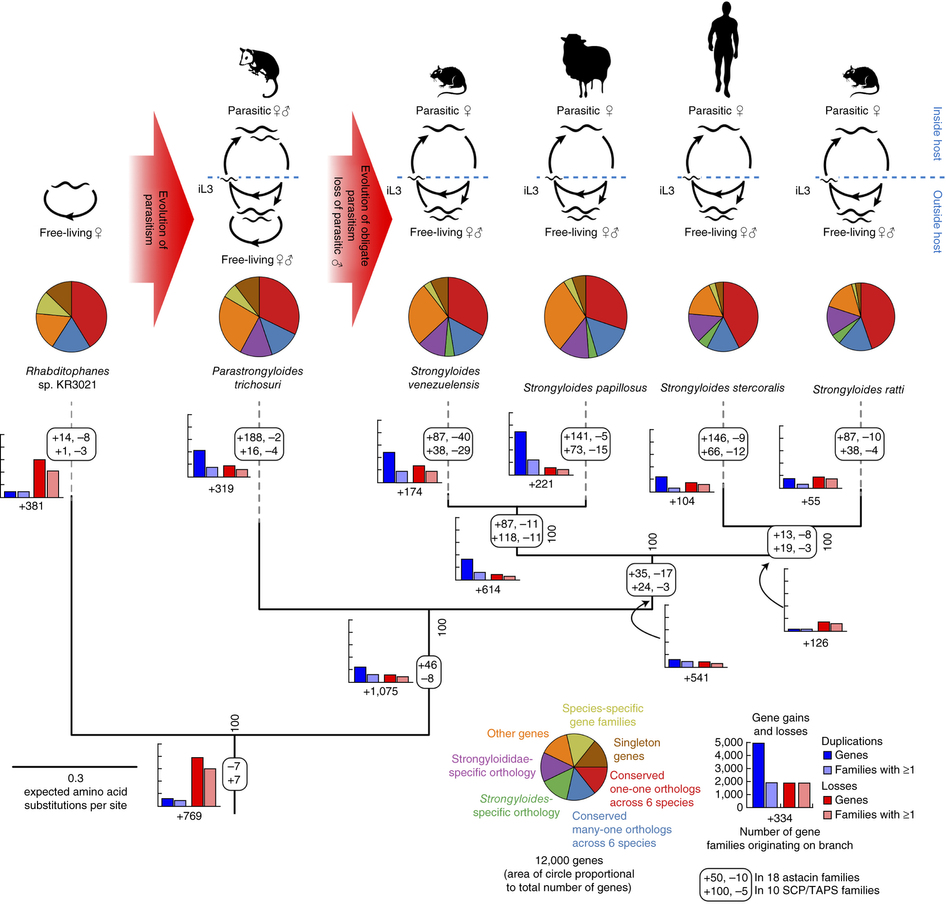
The life cycle of the 6 sequenced species and the gene gains and losses in each lineage. {credit}Hunt et al. Nat. Genet. 2016{/credit}
Two gene families were especially expanded in parasitic genomes—those encoding SCP/TAPS and astacin-domain proteins—and based on RNA-sequencing studies, these were also much more highly expressed in parasitic females than free-living females of the same species. This suggests that these gene families in particular are important for the ability of the worm to infect its host. In support of this hypothesis, the authors found that proteins from these two families are secreted by the worms, and would therefore be able to interact with host tissues to aid in invasion and migration.
Asked about the next steps that need to be taken for these findings, Mark Viney said, “For these SCP/TAPS coding genes what we really need to do is to find out what these genes are doing—this is completely unknown at the moment. For the astacins we can probably guess what they do—being involved in digesting host tissue so that the parasites can feed. They might be potential drug targets.”
The study brought together groups from the UK, Japan, Taiwan, Germany, USA, Mexico and Australia and is one of many examples of successful collaboration in science. “The field of parasitology is a very friendly and interactive community,” said Professor Viney, “so this collaboration was very easy to bring together, and worked extremely well—and will do in the future as well.”
To learn more about this study, check out this blog post from one of the co-first authors, Adam Reid, at the Wellcome Trust Sanger Institute. More coverage can also be found at the University of Bristol website.
Reference:
Hunt, V.L., Tsai I.J., Coghlan, A., Reid, A.J., et al. The genomic basis of parasitism in the Strongyloides clade of nematodes. Nat. Genet. (doi: 10.1038/ng.3495, 1 February 2016)
The paper is available for free online: https://www.nature.com/ng/journal/vaop/ncurrent/full/ng.3495.html