We’ve known for over a century that sensory cortex is arranged in distinct layers, each containing a different make up of neuronal types and projection patterns, but we don’t actually know that much about the actual computations performed in each layer. Today a paper from Massimo Scanziani’s lab takes a big step towards cracking the function of the bottom layer (layer 6) in mice. Layer 6 neurons project both to upper cortical layers and to the lateral geniculate nucleus in the thalamus, which itself is the primary input to cortex, and so are primed to play a large modulatory role. Using a monumental combination of optogenetics, intracellular recording, and behavioral testing, the paper convincingly makes the case that layer 6 controls the gain of visual responses of upper layer neurons (i.e. changes the size of their responses without altering their selectivity). Gain control is a fundamental computation in cortex, and has been invoked as a mechanism for attention, perception, spatial processing, and more. The cellular mechanism here is worked out in primary visual cortex, but it could potentially operate throughout layered cortex.
Category Archives: New in Nature
Awakening dormant genes with cancer drugs
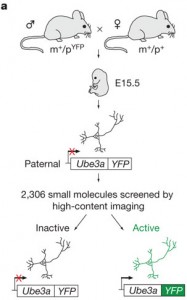
Here’s one that first appeared online at the end of last year by Benjamin Philpot, Bryan Roth and Mark Zylka about a finding that could lead to a therapy for Angelman Syndrome. Angelman syndrome is a rare neurodevelopmental disorder affecting 1 in 15,000 live births and is characterized by developmental delay, lack of speech, seizures, and motor difficulties. There are no therapies available for core symptoms and individuals generally require care throughout life. Autism is often diagnosed in Angelman Syndrome individuals, and the same genomic region has been fingered as a culprit in both disorders. Angelman Syndrome is most commonly caused by deletion of a region on the maternal copy of chromosome 15 containing the gene UBE3A, conversely, some forms of autism may also be caused by duplication of this region.
Although we all possess two copies of UBE3A, only the one inherited from the mother is active. Normally, the paternal copy is epigenetically silenced. This means that in Angelman Syndrome there is no functioning copy at all, which has consequences for multiple signaling pathways and brain circuits. The authors of this paper set out to find a workaround: something that could activate the intact, but dormant, paternal copy of UBE3A. They made a reporter assay from neurons of mice expressing fluorescent paternal UBE3A protein, and performed a large-scale drug screen, testing over 2000 compounds for ones that would activate paternal Ube3A. None of the most likely suspects worked, but an unlikely class of drugs, topoisomerase inhibitors, did so reliably. Even better, one of the best (topotecan) is already an FDA-approved cancer drug.
Patients help bring the study of Alzheimer’s to the dish
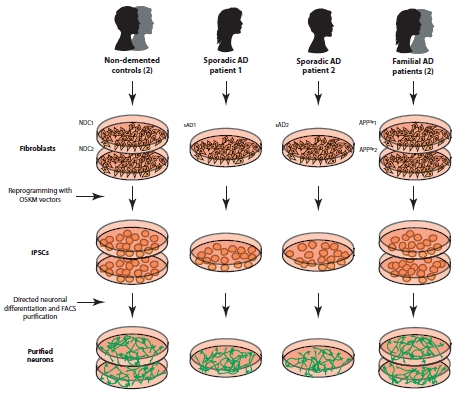
Alzheimer’s disease (AD) is a devastating neurodegenerative disease that could become an even more massive public health problem than it already is, if current projections hold. Some predict that by 2050, 1 in 85 individuals will be affected by the disease. Currently, there is no cure, but there are neurotransmitter-enhancement-based strategies to slow down the cognitive deficits [the loss of cholinergic neurons is implicated in some of the memory problems associated with AD so therefore, pharmacological enhancement of brain acetylcholine concentration can partially alleviate some memory-based symptoms.] However, as with many neurodegenerative diseases, these stop-gap treatments only work for so long, until the cells responding to neurotransmitter supplementation treatments die off completely. Therefore, diverse strategies designed to cure or at least slow down AD are imperative.
While a number of AD transgenic mouse models have been created, based on the various mutations identified in patients, the trouble is that these models still utilize the cross-species approach of studying “diseased” mouse neurons expressing mutated human genes. And perhaps an even bigger problem with many mouse models, genetically-inherited forms of AD represent only ~0.1% of cases, with the remainder being “sporadic” (although there are genetic risk factors influencing the emergence of sporadic AD.)
Timely inhibition
This week’s paper is by Abigail Person and Indira Raman and is about information transmission between two cell populations in the cerebellum – purkinje cells in the cortex and their targets in the deep nuclei. Purkinje cells are justifiably famous for their spectacular anatomy which enables integration of thousands of inputs. This paper, however, is about their output and how these exclusively GABAergic cells control the activity of downstream neurons. Conventional wisdom holds that there should be a straightforward inverse relationship between the firing rate of the two populations, but this has not always been observed. Person and Raman present a new solution based on spike timing – when purkinje cells spike asynchronously, their targets are inhibited (as expected), but when they spike synchronously, nuclear neurons can spike during the gaps in inhibition and end up time locking their activity to their inputs.
This is an intriguing proposal for how information is transmitted in the cerebellum that could have implications for how this brain structure controls movement, but it’s just the first step. The proposal is built from in vitro experiments, deduction, and some supporting in vivo data, but several crucial unknowns have to be resolved before we’ll know whether it’s relevant to actual behavior. There was plenty of spirited discussion during the review process about the strength of some of the authors’ assumptions. There were deeply divided views on whether the authors had made sufficiently strong a case for how the cerebellum IS operating, as opposed to just proposing how it COULD be. We had to decide whether to publish a paper that everyone agreed was interesting, but one that contained some pieces of indirect evidence and some good (but by no means universally agreed-upon) assumptions.
Light dissection of reward
Out online in Nature today: a paper from Naoshige Uchida and colleagues about cell-type specific reward and punishment signals in the ventral tegmental area (VTA) of mice. The VTA is a midbrain region heavily implicated in reward and addiction, and its outputs are thought to provide reward-related signals to other brain areas. One subpopulation of cells with the VTA, the dopaminergic neurons, have been the topic of intense study for their potential computational role in reward learning. Over a decade ago Wolfram Schultz and colleagues found that in monkeys, dopaminergic neurons fired for unexpected rewards, but were also suppressed if expected rewards were not received. Schultz and colleagues proposed that the neurons were representing the difference between expected and actual outcome, and also noted that such reward prediction error has been theoretically posited to drive reinforcement learning. Although reward prediction is by no means the only proposed role for dopamine, the idea that dopaminergic neurons carry reward signals has figured prominently into theories of VTA function and what goes wrong in disease.
But only around half of VTA neurons are dopaminergic; GABAergic neurons, which make inhibitory projections onto dopaminergic neurons, make up a big chunk of the remainder. In the current paper, Uchida and colleagues asked how the two populations encode learned rewards and punishments. They recorded from VTA neurons in mice learning to associate odors with rewards and punishments and sorted the neurons post-hoc by their firing properties. Some neurons had brief phasic responses to rewards and reward-predicting cues. Others had sustained increases in firing during the delay between cues and rewards, and yet others sustained decreases. The authors then used optogenetic stimulation to establish dopaminergic or GABAergic identity in a subset of the cells. Dopaminergic neurons all belonged to the first class of cells with phasic reward and reward-predicting responses, and GABAergic neurons the second class with tonic increases. Most but not all dopaminergic neurons were inhibited by aversive stimuli, most GABAergic neurons were excited. Continue reading