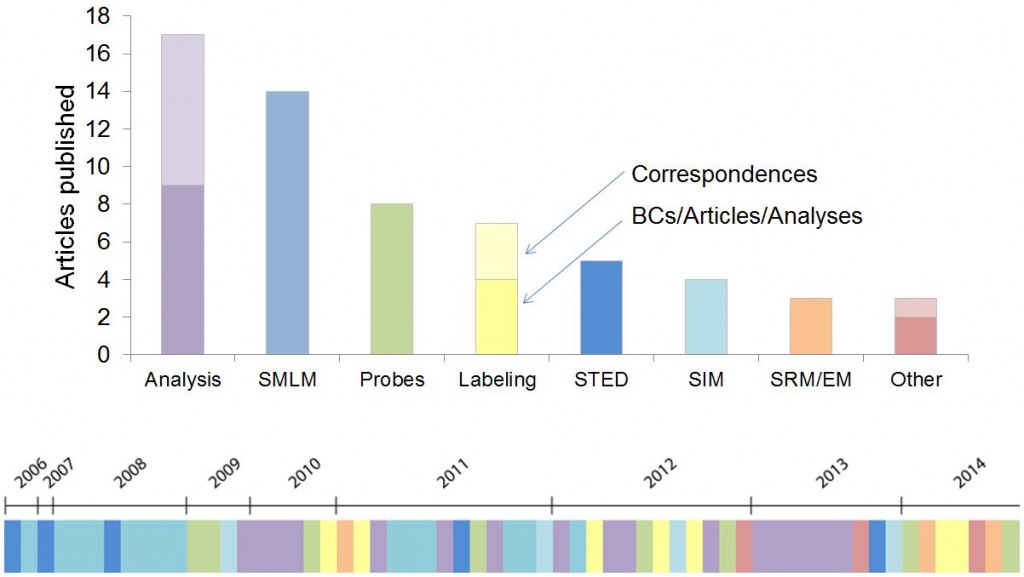
The optogenetic manipulation of cellular properties has not only revolutionized neuroscience, but this technology can also be applied to the manipulation of signaling pathways, transcription or other processes in non-neuronal cells. Here, we highlight some of the papers we have published on the neuroscience side of optogenetics.
Optogenetic tools
2014 has been an exciting year for us with the publication of new optogenetic tools. Klapoetke and Boyden developed Chrimson and Chronos, two channelrhodopsins that they discovered in a screen of algal transcriptomes. Chrimson is more red-shifted than previously known channelrhodopsins while Chronos has faster kinetics. Hochbaum and Cohen described another algal channelrhodopsin called CheRiff, which is highly sensitive to blue light stimulation, making it compatible with red-shifted voltage sensors.
Previously, we published papers describing modifications to optogenetic tools. For example, Prakash and Deisseroth tailored opsin with custom properties. To ensure stoichiometric expression of optogenetic activators and/or inhibitors, Kleinlogel and Bamberg simply and elegantly fused the two proteins into a single chain. Depending on the two partners, this marriage can lead to synergisms or bidirectional effects. Finally, Mattis and Deisseroth undertook a comprehensive characterization of available tools.
Optogenetic applications
Since the initial description of Channelrhodopsin2 (ChR2) as an efficient tool to evoke neural activity in a light-dependent manner, we have seen a flurry of papers applying ChR2 for a variety of questions in neuroscience. For instance, Zhang and Oertner combined this tool with two-photon calcium imaging in rat slices to study synaptic plasticity. Liewald and Gottschalk applied the same methodology to analyze synaptic function in freely moving C. elegans.
ChR2 can also be used to map the function of brains regions as Ayling and Murphy demonstrated by evoking activity in limb muscles via light stimulation in the motor cortex of ChR2 transgenic mice. Similarly, Guo and Ramanathan mapped neural circuitry in C. elegans by combining ChR2-mediated neural activation with imaging of a genetically encoded calcium sensor in downstream neurons. To facilitate circuit mapping in mice, Zhao and Feng generated mouse lines that express ChR2 in GABAergic, cholinergic, serotonergic or parvalbumin-expressing neurons.
While ChR2 is a very popular tool in optogenetics, other family members can do the job as well. C1V1T is a fusion of two different opsins and is particularly useful when applying two-photon excitation, as shown by Packer and Yuste. ReaChR is activated by red light and thus especially useful in vivo. Inagaki and Anderson studied courtship behavior in Drosophila with this tool.
Method of the Year
We celebrated the impact of optogenetics by recognizing the technology as our Method of the Year 2010. We marked the occasion with the publication of special Commentaries on the subjects. Deisseroth discussed the past, present and future of optogenetics. Hegemann and Möglich deliberate on the exploration of new optogenetic tools. And Peron and Svoboda illuminated us on the precise delivery of optogenetic stimulation. In addition, our News Feature recounted the stories behind the “Light tools”.
If we have sparked your interest, the mentioned papers are listed below.
We are excited to hear about the upcoming developments in optogenetics from you.
Nathan C Klapoetke, Yasunobu Murata, Sung Soo Kim, Stefan R Pulver, Amanda Birdsey-Benson, Yong Ku Cho, Tania K Morimoto, Amy S Chuong, Eric J Carpenter, Zhijian Tian, Jun Wang, Yinlong Xie, Zhixiang Yan, Yong Zhang, Brian Y Chow, Barbara Surek, Michael Melkonian, Vivek Jayaraman, Martha Constantine-Paton, Gane Ka-Shu Wong & Edward S Boyden
Independent optical excitation of distinct neural populations
Nature Methods 11, 338–346 (2014) doi:10.1038/nmeth.2836
Daniel R Hochbaum, Yongxin Zhao, Samouil L Farhi, Nathan Klapoetke, Christopher A Werley, Vikrant Kapoor, Peng Zou, Joel M Kralj, Dougal Maclaurin, Niklas Smedemark-Margulies, Jessica L Saulnier, Gabriella L Boulting, Christoph Straub, Yong Ku Cho, Michael Melkonian, Gane Ka-Shu Wong, D Jed Harrison, Venkatesh N Murthy, Bernardo L Sabatini, Edward S Boyden, Robert E Campbell & Adam E Cohen
All-optical electrophysiology in mammalian neurons using engineered microbial rhodopsins
Nature Methods 11, 825–833 (2014) doi:10.1038/nmeth.3000
Rohit Prakash, Ofer Yizhar, Benjamin Grewe, Charu Ramakrishnan, Nancy Wang, Inbal Goshen, Adam M Packer, Darcy S Peterka, Rafael Yuste, Mark J Schnitzer & Karl Deisseroth
Two-photon optogenetic toolbox for fast inhibition, excitation and bistable modulation
Nature Methods 9, 1171–1179 (2012) doi:10.1038/nmeth.2215
Sonja Kleinlogel, Ulrich Terpitz, Barbara Legrum, Deniz Gökbuget, Edward S Boyden, Christian Bamann, Phillip G Wood & Ernst Bamberg
A gene-fusion strategy for stoichiometric and co-localized expression of light-gated membrane proteins
Nature Methods 8, 1083–1088 (2011) doi:10.1038/nmeth.1766
Joanna Mattis, Kay M Tye, Emily A Ferenczi, Charu Ramakrishnan, Daniel J O’Shea, Rohit Prakash, Lisa A Gunaydin, Minsuk Hyun, Lief E Fenno, Viviana Gradinaru, Ofer Yizhar & Karl Deisseroth
Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins
Nature Methods 9, 159–172 (2012) doi:10.1038/nmeth.1808
Yan-Ping Zhang & Thomas G Oertner
Optical induction of synaptic plasticity using a light-sensitive channel
Nature Methods 4, 139 – 141 (2006) doi:10.1038/nmeth988
Jana F Liewald, Martin Brauner, Greg J Stephens, Magali Bouhours, Christian Schultheis, Mei Zhen & Alexander Gottschalk
Optogenetic analysis of synaptic function
Nature Methods 5, 895 – 902 (2008) doi:10.1038/nmeth.1252
Oliver G S Ayling, Thomas C Harrison, Jamie D Boyd, Alexander Goroshkov & Timothy H Murphy
Automated light-based mapping of motor cortex by photoactivation of channelrhodopsin-2 transgenic mice
Nature Methods 6, 219 – 224 (2009) doi:10.1038/nmeth.1303
Zengcai V Guo, Anne C Hart & Sharad Ramanathan
Optical interrogation of neural circuits in Caenorhabditis elegans
Nature Methods 6, 891 – 896 (2009) doi:10.1038/nmeth.1397
Shengli Zhao, Jonathan T Ting, Hisham E Atallah, Li Qiu, Jie Tan, Bernd Gloss, George J Augustine, Karl Deisseroth, Minmin Luo, Ann M Graybiel & Guoping Feng
Cell type–specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function
Nature Methods 8, 745-752 (2011) doi:10.1038/nmeth.1668
Adam M Packer, Darcy S Peterka, Jan J Hirtz, Rohit Prakash, Karl Deisseroth & Rafael Yuste
Two-photon optogenetics of dendritic spines and neural circuits
Nature Methods 9, 1202–1205 (2012) doi:10.1038/nmeth.2249
Hidehiko K Inagaki, Yonil Jung, Eric D Hoopfer, Allan M Wong, Neeli Mishra, John Y Lin, Roger Y Tsien & David J Anderson
Optogenetic control of Drosophila using a red-shifted channelrhodopsin reveals experience-dependent influences on courtship
Nature Methods 11, 325–332 (2014) doi:10.1038/nmeth.2765
Karl Deisseroth
Optogenetics
Nature Methods 8, 26–29 (2011) doi:10.1038/nmeth.f.324
Peter Hegemann & Andreas Möglich
Channelrhodopsin engineering and exploration of new optogenetic tools
Nature Methods 8, 39–42 (2011) doi:10.1038/nmeth.f.327
Simon Peron & Karel Svoboda
From cudgel to scalpel: toward precise neural control with optogenetics
Nature Methods 8, 30–34 (2011) doi:10.1038/nmeth.f.325
Monya Baker
Light tools
Nature Methods 8, 19–22 (2011) doi:10.1038/nmeth.f.322