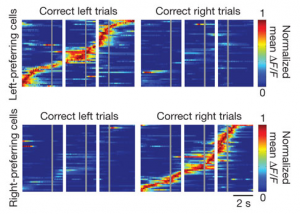
We published another double header yesterday, this time on the role of particular cell types in visual responses. Both studies describe the effect of optogenetically manipulating various interneuron classes in mouse visual cortex. The papers are Lee et al. from Yang Dan‘s lab and Wilson et al. from Mriganka Sur‘s labs. And in fact, both were preceded by Atallah et al. from Massimo Scanziani’s lab, which appeared in Neuron earlier this year. Which means a bonanza of data on the effects of activating parvalbumin-expressing interneurons, and also a bonanza of different conclusions about their exact role – everyone comes to slightly different conclusions.
Tag Archives: neuroscience
A tale of three papers
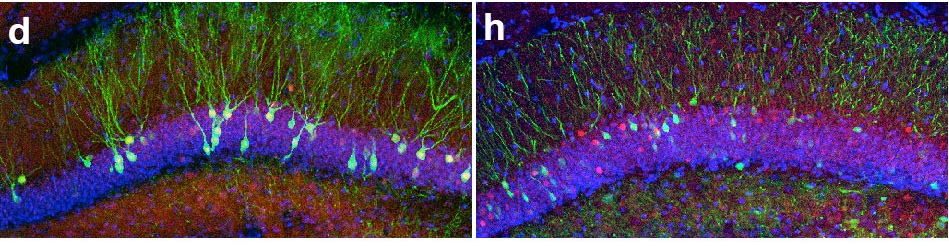
I wanted the title of this post to be “A tale of two one two three papers” but I couldn’t figure out how to get strikethroughs in the title field. And I thought “A tale of two, make that one, no make that two again, oops now three” might be a bit cumbersome. As promised, here’s another installment of the discussion of what happens when we receive conceptually related/overlapping papers. It starts with a paper that appeared just yesterday in Neuron by Kenichi Ohki and colleagues describing how mouse visual cortex neurons that developed from the same neural progenitor cell tend to be more similar functionally than those that did not.
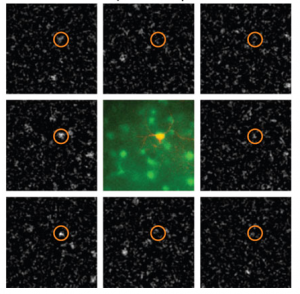
Why is this significant? First a little background. Cells in visual cortex are tuned to different aspects of visual stimuli, such as orientation or direction, and anatomically are organized quite specifically. Cells with similar preferences tend to cluster together and to be selectively connected with each other (though to differing degrees in different species), and this specificity may underlie some of the many computations required to turn photons of light hitting our eyes into comprehensible percepts. It’s been proposed that this clustering could start in early development; neurons born from the same neural progenitor migrate vertically to form columns of sibling neurons, and could be the basis for clusters of adult cells with similar properties. That link hasn’t been demonstrated experimentally until now, and Ohtsuki et al. provides some evidence in support of it.
Now, visual cortex aficionados among you may think this sounds a bit similar to Li et al., a paper by Yang Dan and colleagues that appeared a few months ago, and indeed it is. And you may also recall that THAT paper appeared alongside Yu et al. from Songhai Shi’s lab about the development of synapses between sibling neurons.
So here’s the story from the beginning (or rather, the beginning of our involvement with the manuscripts).
Autism, synapses and mice – pairs division
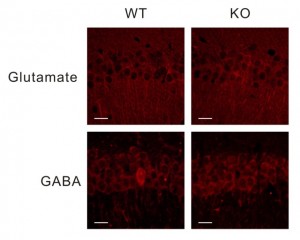
Again, we’re behind on blogging – you guys are keeping us busy with great neuroscience – but here is the story of a pair of papers that appeared back to back in last week’s issue and a continuation of the discussion started here by Noah about the process of joint publication. The two papers by Tobias Boeckers and colleagues and by Eunjoon Kim and colleagues were independently submitted and both describe autism-like phenotypes of mice with mutations in the gene Shank2. In human studies, SHANK2 has been associated with rare cases of autism and these two mice add to the ever-growing list of rodents (according to SFARI.org, 17 rodent models debuted in 2011 alone) that are being created to investigate the functional consequences of genetic mutations linked to autism, in the hopes of understanding mechanisms underlying core symptoms. Shank2 is a scaffolding protein that regulates excitatory synapse function by holding together various molecules such as neurotransmitter receptors and signaling proteins. Mutations in another member of the same gene family, SHANK3, are also associated with human autism, and mutant mice display behaviors reminiscent of ASD symptoms, such as social deficits and obsessive behavior. So this protein family, and more generally, glutamatergic transmission, is potentially one promising line of investigation. Continue reading
Positive feedback drives network (and manuscript) maturation.
It really is an embarrassment of riches here at Nature these days, what with so many excellent neuroscience-related studies emerging. Just in the last couple of weeks, we’ve had the following studies:
- Covert learning by a basal ganglia circuit, despite no participating in the behavioral practice.
- Reach and grasp by people with tetraplegia using a neurally-controlled robotic arm.
- A non-transcriptional circadian cycle conserved across all domains of life.
- Identification of mechanisms linking cerebro-vascular integrity to neurodegeneration.
- Metabolic coupling of glia with axons in the long-term maintenance of axonal integrity.
So really, a lot to write about from a science perspective. However, this blog is dedicated to bringing you the editorial back-story, so I wanted to touch on yet another interesting study, published in print today. This new paper offers an opportunity to discuss an important editorial issue: the manuscript appeal process. For more details, you can always read the appropriate section in our guide to authors. But it’s often helpful to follow a particular [successful] example in order to illustrate the process. Continue reading
“There is no spoon…”: Paralyzed fish navigates virtual environment while we watch its brain
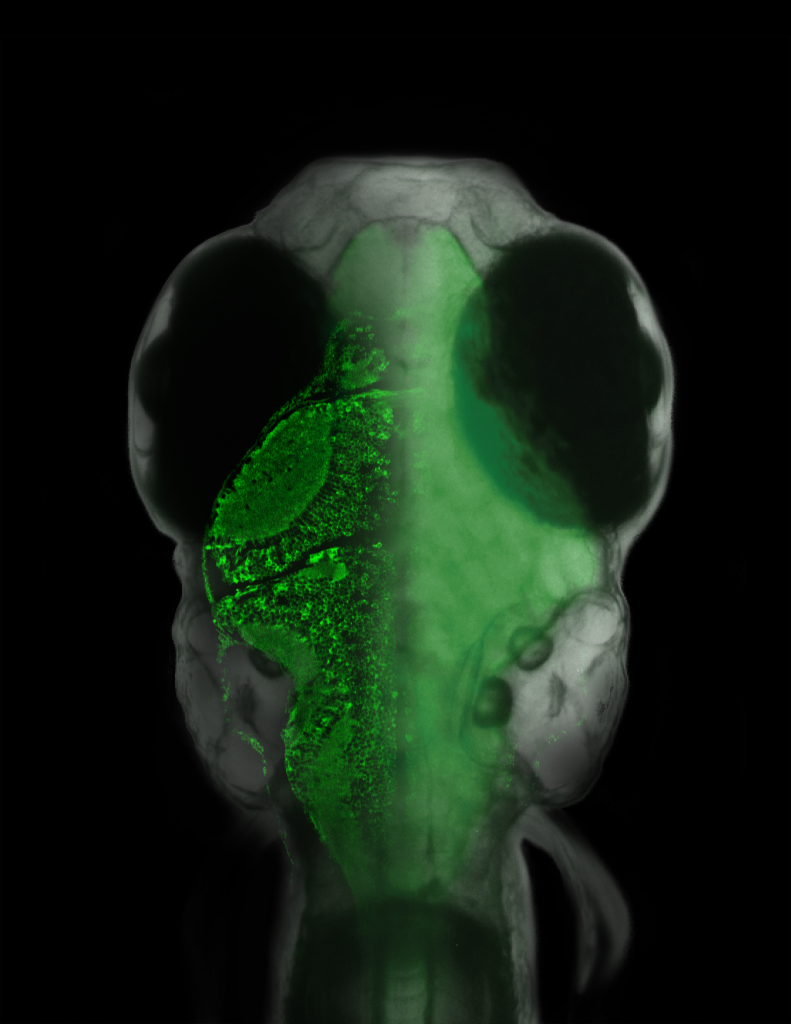
Overlaid on the micrograph of the fish is a slice of its brain measured with a laser scanning microscope, in which single neurons are visible.{credit}(courtesy of Ahrens et al.){/credit}
Sometimes an experiment will just reach off the page and slap you in the face, demanding attention. This happens to me every so often and I must admit, our latest paper from the lab of Florien Engert induced such an experience. There have been several cool, technical tours-de-force (is that proper grammar??) over the last few years involving different creatures navigating in a virtual environment while neuronal activity was monitored. These include a mouse running on a spherical treadmill, as well as a fly marching along a similar treadmill-style ball. But in these examples, having the subject head-fixed (for the stability of recordings in the brain, either with electrodes or through imaging) was moderately non-intrusive since walking motions were independent of the head. The same can’t be said for the subject in this latest example of a virtual reality navigator: a wriggling, swimming fish. Therefore, a more creative solution had to be sought and in a paper published online yesterday, Ahrens, Engert and colleagues decided that paralysis was the way to go in order to follow the neural activity of this navigating fish. Continue reading
Fear of the Light
 **PLEASE SEE UPDATES BELOW**
**PLEASE SEE UPDATES BELOW**
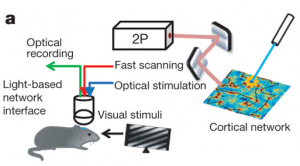
It is commonly believed that distinct mini-networks of neurons, firing together, may be the means by which memories and other conceptual encoding requirements are handled in the brain. However, it is only recently that we have had the tools available to directly test the sufficiency of such a mechanism. Today, a new study in Nature from the lab of Susumu Tonegawa documents the ability to use light as a means to activate distinct subsets of neurons responsible for the encoding of fear memories.
Parietal decision sequences – and more of mice and monkeys
Back in the 1990’s, one of the most intense battlegrounds in systems neuroscience was in monkey posterior parietal cortex. Labs competed to claim what a little strip of cortex called lateral intraparietal area (LIP) really does – decision, movement planning, attention, reward, or all of the above – mostly using single cell recording in behaving monkey. The experiments were (and still are) tough: standard operating procedure requires a well-trained monkey who will perform hundreds if not thousands of trials a day and then isolating neurons one at a time to find ones that respond during some interesting part of the trial. And then lots and lots of repetition so that you can average over many neurons. All things considered, it’s remarkable how much the field has been able to learn with this toolbox.
Fast forward to present day, there’s a new kid on the block. As I discussed a few weeks ago, rodent behavior and physiology is booming. People are taking on questions previously studied mainly in primates and are taking full advantage of the recent storm of new techniques. This is typified by today’s paper by Chris Harvey, Philip Coen, and David Tank, which goes back to the question – what does posterior parietal cortex do during a decision task? They imaged populations of neurons while mice used visual cues to navigate a virtual maze. Just like in primates, individual neurons were selective for different choices that the mouse made. But unlike in primate parietal cortex, where neurons tend to have sustained responses leading up to the time of decision, individual neurons responded transiently in different portions of the trial. So as a population, different choices were represented by distinct sequences of neuronal activity. This kind of sequential firing has been seen in other parts of the rodent brain such as hippocampus, but not in posterior parietal cortex. Continue reading
The Fine Architecture of Learning and Joint Publication
You warily walk into a dark compartment, wondering if there is food inside. Suddenly there is a loud tone and you feel an uncomfortable surge of electricity through your feet. This goes without saying, but it won’t take long before you will learn to be afraid of that tone. However, over time, you hear the tone without the shock, and slowly (foolishly??) accept that the previous connection may no longer hold.
Or perhaps you are extremely motivated to work for food, given that in your home area, nutrition has been sparse and hard to come by. You see millet seeds seemingly just within the reach of your fore-limb. Though not a typical movement for you, you reach for it. In another instance, you find a different type of food that is difficult to handle. However, it is nourishment nonetheless, so you will learn the required motor skills.
SPOILER ALERT: In each of the above cases, you were a mouse the whole time (I know!) But this is a neuroscience blog, not M.Night Shyamalan’s IMDB page, so perhaps we should focus on what was taking place in the brain as each scenario played out. In both of the cases above, learning was occurring, with new information stored away within the appropriate neural connections of particular brain areas. These situations are on display in a pair of new(ish) papers out in Nature, exploring the structural substrates of such learning and identifying patterns underlying the observed structural changes as learning occurred. Continue reading
Layer magic and monkey business
We’ve known for over a century that sensory cortex is arranged in distinct layers, each containing a different make up of neuronal types and projection patterns, but we don’t actually know that much about the actual computations performed in each layer. Today a paper from Massimo Scanziani’s lab takes a big step towards cracking the function of the bottom layer (layer 6) in mice. Layer 6 neurons project both to upper cortical layers and to the lateral geniculate nucleus in the thalamus, which itself is the primary input to cortex, and so are primed to play a large modulatory role. Using a monumental combination of optogenetics, intracellular recording, and behavioral testing, the paper convincingly makes the case that layer 6 controls the gain of visual responses of upper layer neurons (i.e. changes the size of their responses without altering their selectivity). Gain control is a fundamental computation in cortex, and has been invoked as a mechanism for attention, perception, spatial processing, and more. The cellular mechanism here is worked out in primary visual cortex, but it could potentially operate throughout layered cortex.
Telepathy? I think not
There is just something about neural decoding that captures the imagination. Scientists “reading out brain activity” to infer what someone was seeing or doing sounds like the stuff of science fiction. But in practice, with the right dataset and right computer algorithm, it can be done – providing the question you are trying to query the brain is simple enough. But no matter how simple the question, with every paper comes an orgy of stories in the mainstream press about how scientists can eavesdrop on your thoughts or even engage in electronic telepathy. Thereby infuriating scientists and science journalists in droves, sometimes detracting from some very cool work.
Today I’m going back a few years to a paper that typifies this effect, a study from Jack Gallant‘s lab about a model for decoding natural images from fMRI activity in early visual cortex.