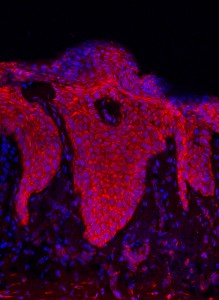
Human psoriatic skin treated with anti-miR-21 {credit}María Jiménez and Juan Guinea-Viniegra{/credit}
Nearly 2% of people worldwide chronically suffer from itchy and painful patches on their bodies, the manifestation of psoriasis, an incurable inflammatory disease in which immune cells infiltrate the skin and release molecules called cytokines that stimulate the skin cells to grow too rapidly. Treatments such as corticosteroids and immunosuppressants can help alleviate mild forms of the disease, and newer antibody-based therapies provide some relief for some of the most severe cases, but some patients fail to respond to these treatments or experience harmful side effects. Now, a new study shows that inhibiting a specific microRNA—a short bit of genetic material that influences the production of proteins in cells—appears to be an effective psoriasis treatment in mice, leaving researchers hopeful that this therapeutic approach will one day be tested in clinical trials.
Psoriasis researchers have known for some time that the levels of a microRNA called miR-21 are elevated in the skin lesions of patients with psoriasis. To determine whether miR-21 plays a crucial role in the disease, a team of scientists led by Erwin Wagner at the Spanish National Cancer Research Centre in Madrid inhibited these genetic elements using an anti-miR-21 treatment. The anti-miR-21 molecules are tiny strands of nucleotides that specifically glom onto miR-21 and prevent it from functioning. Wagner and his colleagues injected this treatment into the skin of mice bearing grafts of diseased tissue from human patients with psoriasis. The anti-miR-21 reduced the thickness of the human skin lesions by about half, a response similar to that obtained using the antibody-based psoriasis therapy etanercept (commercially available from California-based Amgen as Enbrel).