Tuberculosis is an old disease that demands new drugs. More than one million people die each year from Mycobacterium tuberculosis infections and a growing percentage of new infections—at least 9%—are caused by strains of the bacterium that can’t be killed with many of the drugs now available.
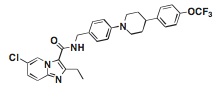
A new experimental compound could help. In a paper published online today in Nature Medicine, researchers describe a small molecule called Q203 that thwarts drug resistant tuberculosis infections in mice by targeting the mycobacterial cytochrome bc1 complex—a mechanism distinct from that of existing agents.
“Q203 works in ways [other] drugs do not,” says Kevin Pethe, project head of the antitubercular program at the Institute Pasteur Korea in Gyeonggi-do, who led the study, “and it can work against the resistant bacteria.”
To find the new drug, Pethe and his colleagues screened more than 100,000 different chemical compounds for their ability to inhibit tuberculosis growth in mouse macrophages. They identified 106 molecules that killed the infectious agent without harming the cells. One compound—a kind of imidazopyridine amide (IPA)—stood out for its ability to wipe out drug-resistant strains of tuberculosis isolated from human clinical specimens. The researchers made small changes in the chemical structure of this molecule to derive Q203. They then tested the compound in mice infected with tuberculosis, and observed that animals given Q203 showed fewer lung lesions than those treated with isoniazid, a commonly used first-line anti-tuberculosis agent. Plus, the mice tolerated high doses of Q203 without any noticeable side effects.
To understand how Q203 stopped the bacteria from replicating, Pethe’s team studied six tuberculosis strains that were resistant to the killing power of Q203. By sequencing the genome of these strains, the researchers pinpointed a common mutation affecting the cytochrome bc1 complex, which is involved in energy metabolism. They then measured ATP levels in Q203-sensitive cells and showed that ATP production dropped significantly after treatment with the experimental agent.
The finding that the cytochrome bc1 complex is the primary target of Q203 is consistent with the results of two recent reports showing that IPAs can broadly inhibit energy transduction systems in the tuberculosis pathogen. In one study, a British team from the University of Birmingham and the pharma giant GlaxoSmithKline discovered a series of molecules in this same chemical class directed at the same target, although these compounds were less effective at inhibiting tuberculosis growth as Q203. In the other report, Pethe and his former colleagues at the Novartis Institute for Tropical Diseases in Singapore showed that a panel of 13 different IPA compounds could kill tuberculosis by depleting ATP.
“The IPAs are getting a lot of attention because they are really inexpensive to make, seem to be safe, and work against drug-resistant tuberculosis,” says Marvin Miller, an organic chemist at the University of Notre Dame in South Bend, Indiana, who, together with colleagues at Indiana’s Eli Lilly, reported earlier this year on yet another set of IPAs with promising anti-tuberculosis and pharmacokinetic properties. “They could be very practical.”