This year marks the 50th anniversary of the US Surgeon General’s first ever report, which implicated smoking as the primary cause of emphysema and other chronic diseases. Despite decades of research, emphysema—a form of chronic obstructive pulmonary disease (COPD), which ranks among the third leading cause of death in the US—remains incurable.
But a new study provides a glimmer of hope. In a paper published online yesterday in the Journal of Clinical Investigation, researchers show that a compound belonging to the class of drugs known as thiazolidinediones (TZDs) can reverse smoking-induced lung damage in mice. What makes the discovery even more intriguing is that TZDs activate a protein called PPAR-gamma, which acts on DNA, and two of these drugs—namely, Takeda’s Actos (pioglitazone) and GlaxoSmithKline’s Avandia (rosiglitazone)—have been used clinically to treat type 2 diabetes.
At first glance, type 2 diabetes and emphysema might appear to have little in common. But in the last decade several studies have suggested that smoking triggers heightened lung inflammatory responses through pathways that are normally held in check by PPAR-gamma, which is perhaps best known for its crucial role in the development of fat cells and regulation of metabolism.
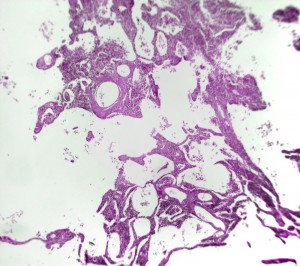
In the new study, led by David Corry and Farrah Kheradmand at Baylor College of Medicine in Houston, the researchers investigated the genetic changes induced by tobacco smoke, and discovered that levels of PPAR-gamma mRNA were depleted in a subset of immune cells from the lungs of smokers with emphysema and mice exposed to cigarette smoke. What’s more, emphysema-associated lung damage began to heal in animals that had ongoing exposure to smoke when they received ciglitazone, an experimental antidiabetic medicine belonging to the TZD class drug that activates PPAR-gamma.